-
E-MAIL
uscert@naver.com

-
E-MAIL
uscert@naver.com

-
TEL
02-529-8005

-
TEL
02-6226-9776


글로벌 RA 인허가
 HOME > 글로벌 RA 인허가 > 시험기술지원
HOME > 글로벌 RA 인허가 > 시험기술지원
시험기술지원
적용분야
의료기기(제조, 수입) 허가 시험
제조 및 수입업체의 자가품질관리를 위한 위탁시험(EO가스 잔류량시험, 무균시험 등)
해외 인증(FDA, CE 인증 등) 및 국제기준규격(IEC)을 위한 시험
유효기간 설정 및 멸균 밸리데이션, 포장 및 클린룸 밸리데이션 등을 위한 시험
관련 규격명적용분야
- 1) IEC 60601-1:2005/A1:2012 Medical electrical equipment - Part 1: General requirements for basic safety and essential performance
- 2) IEC 60601-1-2:2014 Medical electrical equipment - Part 1-2: General requirements for basic safety and essential performance - Collateral Standard: Electromagnetic disturbances - Requirements and tests
- 3) IEC 60601-1-6:2010 Medical electrical equipment - Part 1-6: General requirements for basic safety and essential performance - Collateral standard: Usability
- 4) IEC 60601-1-8:2006 Medical electrical equipment - Part 1-8: General requirements for basic safety and essential performance - Collateral Standard: General requirements, tests and guidance for alarm systems in medical electrical equipment and medical electrical systems
- 5) IEC 60601-1-11:2010 Medical electrical equipment - Part 1-11: General requirements for basic safety and essential performance - Collateral standard: Requirements for medical electrical equipment and medical electrical systems used in the home healthcare environment
- 6) ISO 10993-1:2009 Biological evaluation of medical devices - Part 1: Evaluation and testing within a risk management process
- 7) ISO 10993-5:2009 Biological evaluation of medical devices - Part 5: Tests for in vitro cytotoxicity
- 8) ISO 10993-10:2010 Biological evaluation of medical devices -- Part 10: Tests for irritation and skin sensitization
- 9) ISO 10993-11:2006 Biological evaluation of medical devices - Part 11: Tests for systemic toxicity
관련 규격의 이해
• 전자의료기기
National Standards
 의료기기의 전기 기계적… 의료기기의 전기 기계적… |
 EN 60601-1 EN 60601-1 |
 BS EN 60601-1 BS EN 60601-1 |
 JIS T0601-1 JIS T0601-1 |
 UL 2601-1 UL 2601-1 |
International Standards
| Particular | IEC 60601-2-2 |
| Collateral | IEC 60601-1-2 |
| Base | IEC 60601-1 |
• 의료용품/치과재료
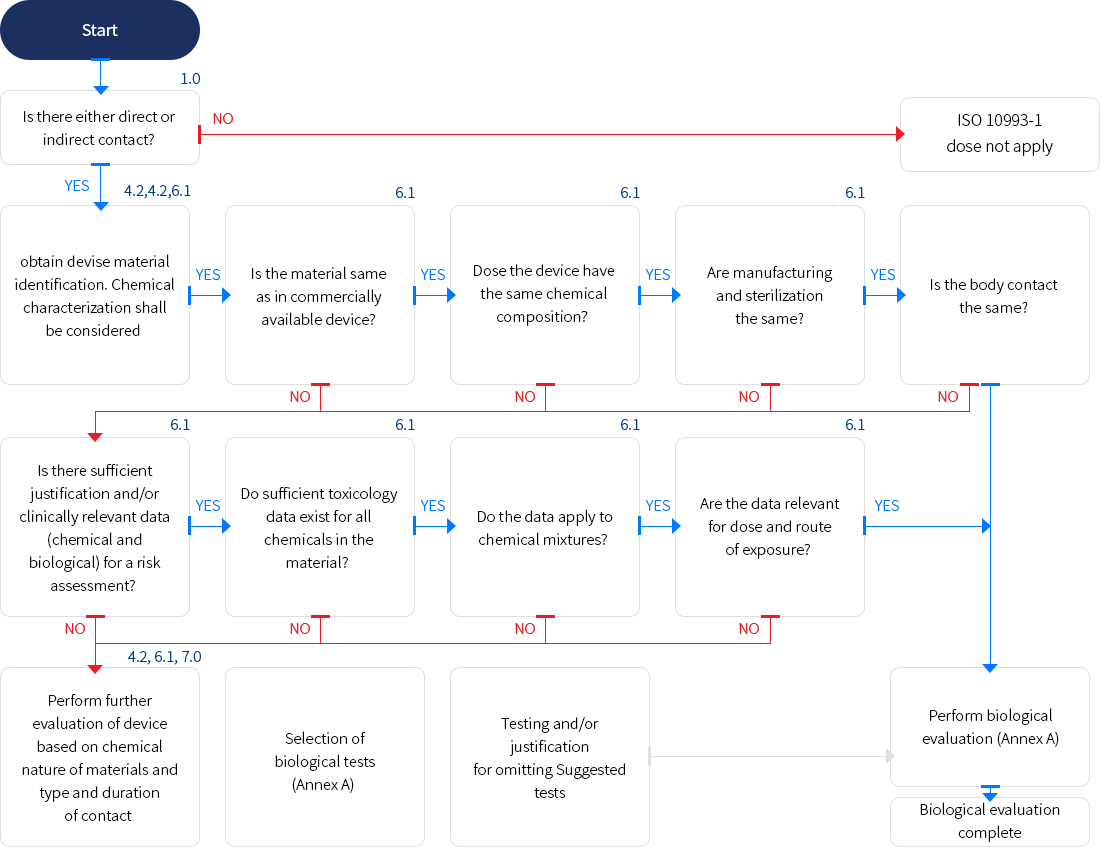
ISO10993-1에 따라 환자의 인체 내 직접 또는 간접적으로 접촉하는 의료기기는 아래의 흐름도를 통하여 ISO10993 적용 여부를 먼저 확인해야 합니다.
※ 위험 관리 프로세스의 일부로 의료 기기의 생물학적 평가에 대한 체계적인 접근 방식 요약
흐름도에 따라 ISO10993 적용여부가 확인 된, 의료용품/치과재료는 아래의 ISO10993-1 Annex A(informative)를 통하여 ISO10993 Test 항목을 확인할 수 있습니다.
| Medical device categorization by | Biological effect | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Nature of body contact (see 5.2) | Concact duration (see5.3) A-limited(≤24h) B-prolongged(>24h to 30d) C-permanent(>30d) |
Cytotoxicith | Sensitization | Irritation or intracutaneous reactivity | Systemic toxicity(acute) | Subchronic toxicity (Subacute toxicity) | genotoxicity | Implantation | Haemocompatibility | |
| Category | Contact | |||||||||
| Surface device | Skin | A | X a | X | X | |||||
| B | X | X | X | |||||||
| C | X | X | X | |||||||
| Mucosal membrane | A | X | X | X | ||||||
| B | X | X | X | |||||||
| C | X | X | X | X | X | |||||
| Breached or compromised surface | A | X | X | X | ||||||
| B | X | X | X | |||||||
| C | X | X | X | X | X | |||||
| External communication device | Blood path, indirect | A | X | X | X | X | X | |||
| B | X | X | X | X | X | |||||
| C | X | X | X | X | X | X | ||||
| Tissue/bone/denitin | A | X | X | X | ||||||
| B | X | X | X | X | X | X | X | |||
| C | X | X | X | X | X | X | X | |||
| Circulation blood | A | X | X | X | X | X | ||||
| B | X | X | X | X | X | X | X | X | ||
| C | X | X | X | X | X | X | X | |||
| Implant device | Tissue/bone | A | X | X | X | |||||
| B | X | X | X | X | X | X | X | |||
| C | X | X | X | X | X | X | X | |||
| blood | A | X | X | X | X | X | X | X | ||
| B | X | X | X | X | X | X | X | X | ||
| C | X | X | X | X | X | X | X | X | ||


 KOR
KOR  ENG
ENG  JPN
JPN  CHN
CHN